Critical Quality Attributes (CQA) are fundamental elements in pharmaceutical development and manufacturing that directly impact product quality, safety, and efficacy. In the modern pharmaceutical industry, ensuring consistent product quality is not optional—it is a regulatory and ethical requirement. This is where Critical Quality Attributes (CQA) play a central role. They act as key indicators that help manufacturers maintain control over product performance and minimize variability during production.
Critical Quality Attributes (CQA) refer to the physical, chemical, biological, or microbiological properties that must be controlled within predefined limits to ensure the desired product quality. These attributes are identified during product development and are continuously monitored throughout the product lifecycle. Proper identification of (CQA) allows for better risk management and more robust manufacturing processes.
Understanding (CQA) is essential for implementing Quality by Design (QbD), maintaining compliance with regulatory guidelines, and ensuring patient safety. Additionally, they support continuous improvement initiatives and help organizations achieve consistent batch-to-batch manufacturing quality, which is critical for building trust with regulatory authorities and patients.

What are Critical Quality Attributes (CQA)?
Critical Quality Attributes (CQA) are measurable characteristics of a drug product that determine its overall quality. These attributes must remain within specific limits to guarantee that the product performs as intended.
Key Characteristics of Critical Quality Attributes (CQA):
- Directly impact product quality
- Linked to safety and efficacy
- Measurable and testable
- Defined during product development
- Controlled throughout manufacturing
For example, in a tablet formulation, Critical Quality Attributes (CQA) may include:
- Assay (drug content)
- Dissolution rate
- Hardness
- Impurity levels
- Moisture content
Each of these Critical Quality Attributes (CQA) must be maintained within acceptable limits.
Importance of Critical Quality Attributes (CQA)
Critical Quality Attributes (CQA) are crucial for ensuring that pharmaceutical products consistently meet regulatory standards and patient expectations. They serve as the backbone of product quality, helping manufacturers maintain control over critical characteristics throughout the product lifecycle. By focusing on Critical Quality Attributes (CQA), companies can reduce variability, improve process understanding, and ensure reliable product performance.
1. Ensures Product Safety
Critical Quality Attributes (CQA) help ensure that the product is safe for human consumption by controlling harmful impurities, contamination, and variability in composition. Maintaining these attributes within defined limits protects patients from potential risks.
2. Maintains Product Efficacy
By controlling Critical Quality Attributes (CQA), manufacturers ensure the drug performs as intended. Attributes such as dissolution, potency, and bioavailability directly impact therapeutic effectiveness.
3. Regulatory Compliance
Regulatory authorities require strict monitoring and control of Critical Quality Attributes (CQA). Proper documentation and adherence to guidelines help organizations meet global regulatory expectations and avoid compliance issues.
4. Supports Quality by Design (QbD)
Critical Quality Attributes (CQA) are the foundation of Quality by Design (QbD). They guide the development process, enabling a scientific and risk-based approach to product and process design.
5. Reduces Product Failures
Proper control of Critical Quality Attributes (CQA) minimizes batch failures, deviations, and product recalls. This not only saves cost but also protects the company’s reputation.
6. Enhances Process Understanding
Focusing on Critical Quality Attributes (CQA) improves understanding of how materials and process parameters affect product quality, leading to better decision-making.
7. Builds Patient and Market Trust
Consistent control of Critical Quality Attributes (CQA) ensures high-quality products, which helps build trust among patients, healthcare professionals, and regulatory bodies.
Types of Critical Quality Attributes (CQA)
Critical Quality Attributes (CQA) can be categorized based on their nature:
1. Physical Attributes
These include properties like:
- Particle size
- Tablet hardness
- Appearance
2. Chemical Attributes
Critical Quality Attributes (CQA) in this category include:
- Assay
- Impurities
- Degradation products
3. Biological Attributes
Applicable for biologics:
- Protein structure
- Bioactivity
- Immunogenicity
4. Microbiological Attributes
These include:
- Sterility
- Microbial limits
- Endotoxins
Examples of Critical Quality Attributes (CQA)
Understanding examples helps clarify the concept of Critical Quality Attributes (CQA).
Example 1: Tablet Dosage Form
- Assay: Ensures correct drug content
- Dissolution: Ensures proper drug release
- Hardness: Affects mechanical strength
Example 2: Injectable Product
- Sterility: Must be free from microorganisms
- pH: Affects stability and compatibility
- Particulate matter: Must be within limits
Example 3: Biopharmaceutical Product
- Protein aggregation
- Glycosylation patterns
- Biological activity
All these are Critical Quality Attributes (CQA) that must be controlled.
How to Identify Critical Quality Attributes (CQA)
Identifying Critical Quality Attributes (CQA) is a systematic and science-based process that ensures all key product characteristics affecting quality, safety, and efficacy are properly defined and controlled. This process is essential for successful pharmaceutical development and regulatory compliance.
Step 1: Define Quality Target Product Profile (QTPP)
The Quality Target Product Profile (QTPP) outlines the desired characteristics of the final product, such as dosage form, strength, route of administration, and stability. It serves as the foundation for identifying Critical Quality Attributes (CQA) by clearly defining what the product is expected to achieve.
Step 2: Perform Risk Assessment
Risk assessment is used to evaluate which attributes have a significant impact on product quality. Common tools include:
- Failure Mode and Effects Analysis (FMEA)
- Risk ranking and filtering
- Hazard analysis
These tools help prioritize potential Critical Quality Attributes (CQA) based on their severity, occurrence, and detectability.
Step 3: Link to Safety and Efficacy
At this stage, each attribute is evaluated to determine its direct impact on patient safety and therapeutic efficacy. Only those attributes that significantly influence clinical performance are classified as Critical Quality Attributes (CQA).
Step 4: Conduct Experimental Studies
Scientific studies and experiments, such as Design of Experiments (DoE), are conducted to confirm the criticality of identified attributes. This step provides data-driven evidence to support the selection of Critical Quality Attributes (CQA).
Step 5: Finalize and Control Critical Quality Attributes (CQA)
Once identified, Critical Quality Attributes (CQA) are documented and integrated into the control strategy. They are continuously monitored and maintained within predefined limits throughout the product lifecycle to ensure consistent quality.
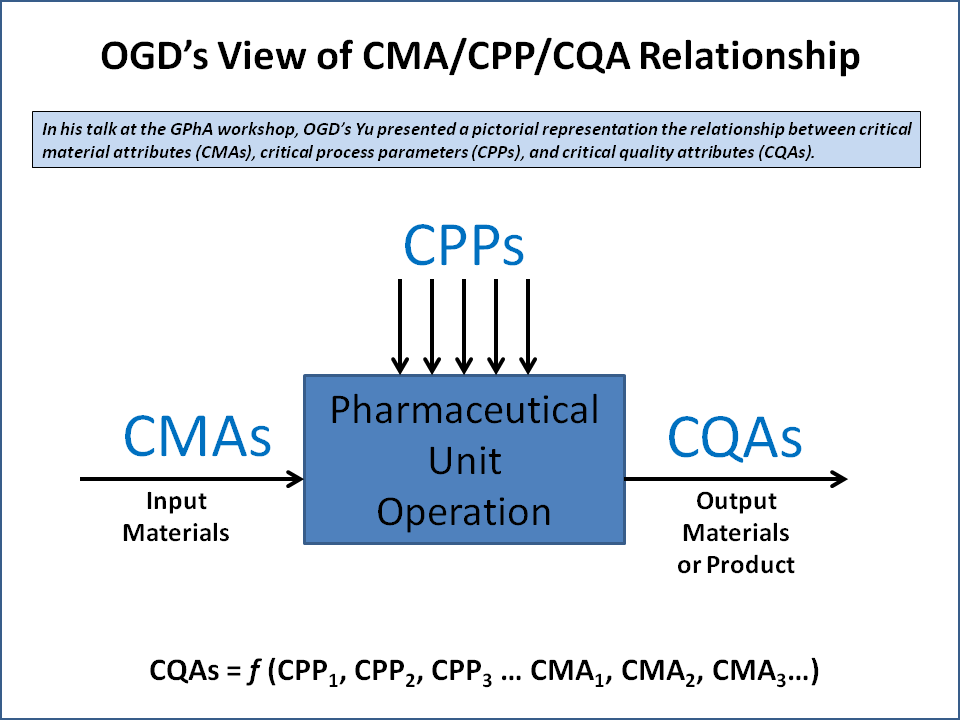
Relationship Between CQA, CPP, and CMA
Critical Quality Attributes (CQA) are closely interconnected with Critical Process Parameters (CPP) and Critical Material Attributes (CMA). Together, these three elements form the foundation of a robust pharmaceutical development and manufacturing system under the Quality by Design (QbD) framework.
Critical Process Parameters (CPP)
Critical Process Parameters (CPP) are the key variables in the manufacturing process that have a direct impact on Critical Quality Attributes (CQA). These parameters must be carefully monitored and controlled to ensure consistent product quality. Examples of CPP include temperature, pressure, mixing speed, and processing time.
Critical Material Attributes (CMA)
Critical Material Attributes (CMA) refer to the physical, chemical, or biological properties of raw materials and excipients that can influence Critical Quality Attributes (CQA). Variations in material properties such as particle size, moisture content, and purity can significantly affect the final product quality.
Relationship Overview
The relationship between these elements can be summarized as:
CMA → CPP → CQA
- Changes in Critical Material Attributes (CMA) can impact how the process behaves.
- Variations in Critical Process Parameters (CPP) can directly affect Critical Quality Attributes (CQA).
- Ultimately, both CMA and CPP must be controlled to maintain Critical Quality Attributes (CQA) within acceptable limits.
Integrated Understanding
A thorough understanding of the relationship between CMA, CPP, and Critical Quality Attributes (CQA) allows manufacturers to develop a strong control strategy. By managing material variability and optimizing process conditions, organizations can ensure consistent product quality, reduce risks, and improve overall manufacturing efficiency.
Role of Critical Quality Attributes (CQA) in Quality by Design (QbD)
Quality by Design (QbD) is a systematic approach to pharmaceutical development. Critical Quality Attributes (CQA) are the backbone of QbD.
Key Roles:
- Define product quality targets
- Guide process design
- Enable risk-based decision making
- Support continuous improvement
Without identifying Critical Quality Attributes (CQA), QbD cannot be effectively implemented.
Regulatory Perspective on Critical Quality Attributes (CQA)
Regulatory agencies emphasize the importance of Critical Quality Attributes (CQA).
Guidelines Include:
- ICH Q8 (Pharmaceutical Development)
- ICH Q9 (Quality Risk Management)
- ICH Q10 (Pharmaceutical Quality System)
Regulators expect:
- Clear identification of Critical Quality Attributes (CQA)
- Justification based on scientific data
- Continuous monitoring and control
Tools Used for Managing Critical Quality Attributes (CQA)
Several tools help in identifying and controlling Critical Quality Attributes (CQA):
1. Risk Assessment Tools
- FMEA
- HACCP
2. Statistical Tools
- Design of Experiments (DoE)
- Process Capability Analysis
3. Analytical Techniques
- HPLC
- GC
- Spectroscopy
4. Process Analytical Technology (PAT)
Real-time monitoring of Critical Quality Attributes (CQA)
Challenges in Managing Critical Quality Attributes (CQA)
Despite their importance, managing CQA can be challenging.
Common Challenges:
- Incorrect identification
- Lack of data
- Process validation
- Regulatory complexity
Solutions:
- Use scientific approaches
- Implement robust risk assessment
- Invest in advanced analytics
- Continuous training
Lifecycle Management of Critical Quality Attributes (CQA)
Critical Quality Attributes (CQA) are not static—they evolve over time as more knowledge is gained about the product and process. Effective lifecycle management ensures that these attributes are consistently controlled from initial development through commercial production and ongoing improvement. This approach aligns with regulatory expectations and supports continuous product quality enhancement.
Stage 1: Development
During the development stage, Critical Quality Attributes (CQA) are identified and defined based on the Quality Target Product Profile (QTPP) and risk assessment studies. Scientific data, prior knowledge, and experimental studies such as Design of Experiments (DoE) are used to determine which attributes are critical. This stage lays the foundation for building a robust control strategy.
Stage 2: Commercial Manufacturing
In the commercial manufacturing stage, (CQA) are routinely monitored and controlled to ensure consistent product quality. Established control strategies, in-process controls, and quality testing are implemented to maintain these attributes within predefined limits. Any variation is carefully evaluated to prevent impact on product quality.
Stage 3: Continuous Improvement
In this stage, ongoing data from manufacturing, quality control, and stability studies are analyzed to further optimize processes. Continuous monitoring of (CQA) enables identification of trends, process improvements, and opportunities for innovation. This ensures sustained product quality and operational excellence over time.
Integrated Lifecycle Approach
Managing (CQA) across the entire lifecycle helps organizations maintain compliance, improve efficiency, and adapt to changes. It also supports a proactive quality culture, where continuous learning and improvement are key to long-term success.
Future Trends in Critical Quality Attributes (CQA)
The future of (CQA) is driven by technology.
Emerging Trends:
- Artificial Intelligence (AI)
- Machine Learning (ML)
- Real-time release testing
- Digital twins
These advancements will enhance control over Critical Quality Attributes (CQA).
Best Practices for Managing Critical Quality Attributes (CQA)
To effectively manage Critical Quality Attributes (CQA), follow these best practices:
- Clearly define QTPP
- Perform thorough risk assessments
- Use data-driven approaches
- Implement real-time monitoring
- Ensure cross-functional collaboration
Conclusion
Critical Quality Attributes (CQA) are the foundation of pharmaceutical quality, playing a vital role in ensuring that products are safe, effective, and compliant with regulatory standards. They provide a scientific framework for understanding and controlling the factors that directly impact product performance. By properly identifying, controlling, and continuously monitoring pharmaceutical companies can achieve consistent product quality and reduce variability across batches.
Moreover, a strong focus on (CQA) supports risk-based decision-making, enhances process understanding, and aligns with modern regulatory expectations such as Quality by Design (QbD). This not only improves operational efficiency but also minimizes the chances of product failures, deviations, and recalls.
In an increasingly competitive and highly regulated environment, mastering (CQA) is not just important—it is essential for long-term success, regulatory compliance, and maintaining patient trust.
Frequently Asked Questions (FAQs)
1. What are Critical Quality Attributes (CQA)?
Critical Quality Attributes (CQA) are measurable properties that determine the quality, safety, and efficacy of a pharmaceutical product.
2. Why are Critical Quality Attributes (CQA) important?
Critical Quality Attributes (CQA) ensure product consistency, regulatory compliance, and patient safety.
3. How are Critical Quality Attributes (CQA) identified?
They are identified through QTPP definition, risk assessment, and experimental studies.
4. What is the difference between CQA and CPP?
Critical Quality Attributes (CQA) are product properties, while CPP are process parameters affecting those properties.
5. Can Critical Quality Attributes (CQA) change over time?
Yes, Critical Quality Attributes (CQA) may evolve based on new data and process improvements.
6. What role do Critical Quality Attributes (CQA) play in QbD?
They form the basis for designing and controlling pharmaceutical processes.
7. Are Critical Quality Attributes (CQA) required by regulators?
Yes, regulatory agencies require identification and control of Critical Quality Attributes (CQA).