Technology transfer in pharmaceuticals is a critical process that ensures the successful transition of a drug product from research and development to commercial manufacturing. In today’s competitive pharmaceutical industry, the ability to transfer knowledge, processes, and technologies efficiently plays a vital role in maintaining product quality and reducing time to market.
Technology transfer in pharmaceuticals involves the systematic sharing of technical knowledge, data, and manufacturing processes between different departments or organizations. This ensures that a product developed in a laboratory environment can be reproduced consistently on a commercial scale. Without effective technology transfer, pharmaceutical companies may face issues such as product variability, delays, and regulatory challenges.
What is Technology Transfer in Pharmaceuticals?
Technology transfer in pharmaceuticals refers to the process of transferring scientific knowledge, manufacturing processes, analytical methods, and quality controls from one unit to another. This typically occurs between research and development (R&D), pilot plant, and commercial manufacturing units.
The primary goal of technology transfer in pharmaceuticals is to ensure that the receiving unit can produce the product with the same quality, safety, and efficacy as developed in the original unit. This involves detailed documentation, training, and validation of processes to ensure consistency across all stages of production.

Importance of Technology Transfer in Pharmaceuticals
Technology transfer in pharmaceuticals is essential for ensuring seamless production and regulatory compliance. It acts as a bridge between product development and large-scale manufacturing.
One of the key benefits of technology transfer in pharmaceuticals is the reduction of risks associated with scaling up production. By transferring accurate data and process knowledge, companies can avoid errors and ensure consistent product quality. Additionally, it helps in reducing time to market, which is crucial in a highly competitive industry.
Furthermore, regulatory authorities require proper documentation of technology transfer activities to ensure compliance with Good Manufacturing Practices (GMP). Therefore, an effective technology transfer process is not only beneficial but also mandatory for pharmaceutical companies.
Objectives of Technology Transfer
The objectives of technology transfer in pharmaceuticals are centered around ensuring consistency, efficiency, and compliance.
The primary objective is to transfer all necessary information required for manufacturing a product. This includes formulation details, process parameters, equipment specifications, and analytical methods. Another important objective is to ensure that the receiving unit can replicate the process without any deviation.
Technology transfer in pharmaceuticals also aims to minimize variability, reduce production costs, and improve overall process efficiency. It ensures that the product meets all quality standards and regulatory requirements.
Types of Technology Transfer in Pharmaceuticals
Technology transfer in pharmaceuticals can be classified into different types based on the direction and purpose of transfer.
1. Vertical Technology Transfer
This involves transferring technology from research and development to production units. It is the most common type of technology transfer in pharmaceuticals.
2. Horizontal Technology Transfer
In this type, technology is transferred between different manufacturing sites or companies. This is often done for contract manufacturing or global production.
3. Intra-Company Transfer
This occurs within the same organization between different departments or locations.
4. Inter-Company Transfer
This involves transferring technology between different companies, such as partnerships or licensing agreements.
Stages of Technology Transfer in Pharmaceuticals
Technology transfer in pharmaceuticals follows a structured and systematic approach to ensure smooth and successful implementation across different stages of manufacturing. Each stage is designed to transfer critical knowledge, processes, and controls from the development unit to the production unit without affecting product quality. This structured process helps in identifying potential risks early and ensures consistency in product performance. Proper execution of each stage is essential to meet regulatory requirements and achieve efficient large-scale-up production.
1. Development Stage
In the development stage, the product is formulated and tested in the laboratory. Detailed information about formulation, process parameters, and analytical methods is generated.
Technology transfer in pharmaceuticals begins here, as proper documentation is created to facilitate future transfer activities.
2. Scale-Up Stage
During the scale-up stage, the process is tested on a larger scale to evaluate its feasibility for commercial production. This stage helps in identifying potential challenges and optimizing process parameters.
Technology transfer in pharmaceuticals plays a key role in ensuring that the process remains consistent during scaling up.
3. Transfer Stage
In this stage, all necessary information is transferred to the manufacturing unit. This includes process instructions, batch manufacturing records, and quality specifications.
Training is provided to the receiving team to ensure proper understanding of the process. Technology transfer in pharmaceuticals ensures that all critical aspects are communicated effectively.
4. Validation Stage
Validation is carried out to confirm that the transferred process works as intended. This involves performing validation batches and analyzing results.
Technology transfer in pharmaceuticals ensures that the process meets all quality and regulatory requirements before commercial production.
5. Commercial Production
After successful validation, the process is implemented for full-scale manufacturing. Continuous monitoring is carried out to maintain product quality.
Technology transfer in pharmaceuticals ensures that the product is consistently produced according to predefined specifications.
Key Elements of Technology Transfer
Key elements of technology transfer in pharmaceuticals include several critical factors that ensure a smooth and successful transition from development to manufacturing. These elements involve detailed documentation, proper equipment selection, clearly defined process parameters, and effective communication between sending and receiving units. Each element plays an important role in maintaining product quality, consistency, and regulatory compliance. By focusing on these key aspects, pharmaceutical companies can minimize risks, avoid process failures, and ensure efficient large-scale production.
1. Documentation
Comprehensive documentation is essential for successful technology transfer. It includes process descriptions, batch records, and analytical methods.
2. Equipment Selection
Selecting appropriate equipment is crucial for replicating the process at a larger scale.
3. Process Parameters
Critical parameters such as temperature, pressure, and mixing speed must be controlled.
4. Training
Proper training ensures that the receiving team understands the process.
5. Quality Assurance
QA ensures that all activities comply with regulatory standards.
Challenges in Technology Transfer
Technology transfer in pharmaceuticals is a complex process and involves several challenges.
One major challenge is maintaining product consistency during transfer. Differences in equipment and environmental conditions can affect the process. Another challenge is inadequate documentation, which can lead to errors and delays.
Communication gaps between teams can also hinder effective technology transfer. Additionally, regulatory requirements add complexity to the process, requiring thorough validation and documentation.
Best Practices for Successful Technology Transfer
To ensure successful technology transfer in pharmaceuticals, companies should follow best practices.
- Develop detailed documentation
- Ensure cross-functional collaboration
- Conduct thorough risk assessments
- Provide proper training
- Use advanced monitoring tools
These practices help in minimizing risks and ensuring smooth transfer of technology.
Process of Technology Transfer in Pharmaceuticals
Technology transfer in pharmaceuticals follows a structured process that includes multiple stages to ensure smooth transition from innovation to commercial production.
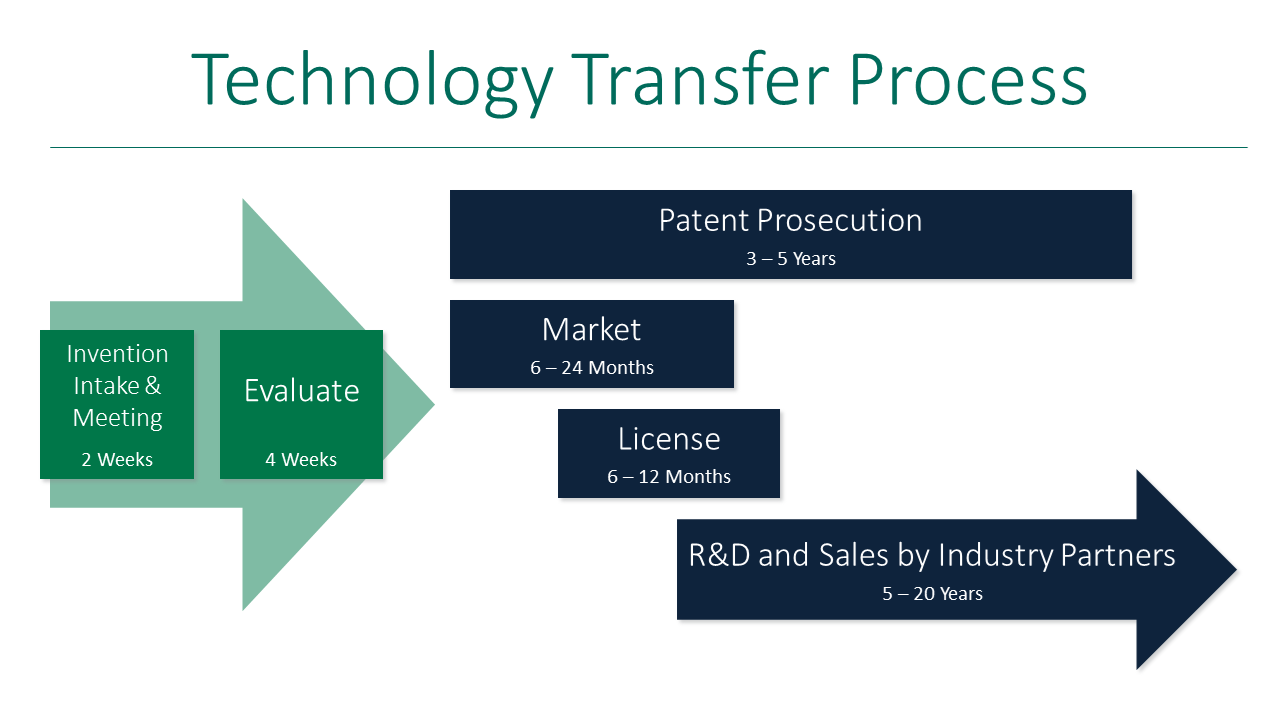
1. Invention Intake & Meeting (2 Weeks)
The process of technology transfer in pharmaceuticals begins with the invention intake and initial meeting stage. In this phase, the developed product or technology is introduced to the concerned teams, and all relevant information is shared. Discussions are carried out to understand the process, objectives, and feasibility. This stage ensures proper communication between research and manufacturing teams, which is essential for successful technology transfer in pharmaceuticals.
2. Evaluation Stage (4 Weeks)
In the evaluation stage, the technical, commercial, and regulatory feasibility of the product is analyzed in detail. Experts review process parameters, scalability, cost, and potential risks involved in production. This stage plays a critical role in technology transfer in pharmaceuticals as it determines whether the product is ready for further development and transfer to the manufacturing unit.
3. Patent Prosecution (3–5 Years)
Patent prosecution is an important stage in technology transfer in pharmaceuticals where intellectual property rights are secured. During this phase, legal procedures are followed to protect the innovation from unauthorized use. Securing patents ensures that the company has exclusive rights to manufacture and commercialize the product, making this step crucial for long-term business success.
4. Market Assessment (6–24 Months)
After securing intellectual property, the product undergoes market assessment to evaluate its commercial potential. In this stage, demand, competition, pricing, and market trends are analyzed. Technology transfer in pharmaceuticals relies on this step to ensure that the product has strong market viability before large-scale investment and production.
5. Licensing Stage (6–12 Months)
In the licensing stage, agreements are made to transfer the technology to manufacturing partners or other organizations. This includes defining roles, responsibilities, and usage rights. Proper documentation and legal agreements are prepared to ensure a smooth and compliant technology transfer in pharmaceuticals.
6. R&D and Commercial Production by Industry Partners (5–20 Years)
The final stage of technology transfer in pharmaceuticals involves large-scale manufacturing, research support, and product commercialization by industry partners. Continuous monitoring, process optimization, and quality control are carried out to maintain product consistency. This stage ensures that the product reaches the market successfully and performs effectively throughout its lifecycle.
Regulatory Considerations
Regulatory compliance is a critical aspect of technology transfer in pharmaceuticals. Regulatory authorities require detailed documentation of the transfer process to ensure product quality and safety.
Companies must follow guidelines such as Good Manufacturing Practices (GMP) and provide evidence that the transferred process produces consistent results. Proper validation and risk assessment are essential for meeting regulatory requirements.
Role of Quality Assurance in Technology Transfer
Quality assurance plays a vital role in technology transfer in pharmaceuticals. QA teams ensure that all processes comply with regulatory standards and internal quality requirements.
They review documentation, approve protocols, and monitor validation activities. Technology transfer in pharmaceuticals relies heavily on QA to ensure consistency and compliance.
Applications of Technology Transfer in Pharmaceuticals
Technology transfer in pharmaceuticals is widely used in various areas of drug development and manufacturing.
It is used in the production of tablets, capsules, injectables, and APIs. It is also essential for contract manufacturing and global product distribution. Additionally, technology transfer is used in biotechnology processes such as fermentation and cell culture.
Future Trends in Technology Transfer
The future of technology transfer in pharmaceuticals is driven by innovation and digital transformation.
Technologies such as artificial intelligence and digital twins are being used to improve process understanding and reduce risks. Continuous manufacturing is also simplifying technology transfer by reducing scale-up challenges.
These advancements are making technology transfer more efficient and reliable.
Conclusion
Technology transfer in pharmaceuticals is a crucial process that ensures the successful transition of products from development to commercial production. It plays a key role in maintaining product quality, reducing risks, and ensuring regulatory compliance.
With proper planning, documentation, and collaboration, pharmaceutical companies can achieve efficient technology transfer and improve overall manufacturing performance.
Frequently Asked Questions (FAQ)
1. What is technology transfer in pharmaceuticals?
It is the process of transferring knowledge, processes, and methods from development to manufacturing.
2. Why is technology transfer important?
It ensures consistent product quality and smooth production.
3. What are the stages of technology transfer?
Development, scale-up, transfer, validation, and commercial production.
4. What are the challenges in technology transfer?
Maintaining consistency, documentation issues, and regulatory compliance.
5. What is the role of QA in technology transfer?
QA ensures compliance with quality and regulatory standards.
6. What are types of technology transfer?
Vertical, horizontal, intra-company, and inter-company.