The pharmaceutical industry is rapidly evolving toward more sustainable, efficient, and biologically driven production methods. One of the most transformative approaches is Fermentation-Based API manufacturing. This advanced biotechnology process uses microorganisms such as bacteria, yeast, and fungi to produce Active Pharmaceutical Ingredients (APIs) that are critical for life-saving medicines.
From antibiotics and vaccines to hormones and enzymes, fermentation has revolutionized pharmaceutical manufacturing. Unlike purely chemical synthesis, fermentation-based production leverages biological systems to create complex molecules with high specificity and lower environmental impact.
What is Fermentation-Based API?
A Fermentation-Based API is an Active Pharmaceutical Ingredient produced through microbial fermentation rather than traditional chemical synthesis. Microorganisms are cultured in controlled environments where they metabolize nutrients and generate desired pharmaceutical compounds.
Common microorganisms used include:
Bacteria (e.g., Streptomyces)
Yeast (e.g., Saccharomyces cerevisiae)
Fungi (e.g., Penicillium species)
Historically, fermentation transformed medicine with the discovery of antibiotics like:
Penicillin
Streptomycin
Erythromycin
Today, fermentation is also widely used in insulin, vaccines, enzymes, and monoclonal antibody production.

History of Fermentation in Pharmaceuticals
The pharmaceutical fermentation revolution began with Alexander Fleming in 1928, when he discovered that Penicillium mold inhibited bacterial growth.
During World War II, industrial-scale fermentation was developed to mass-produce Penicillin. This breakthrough marked the beginning of modern biopharmaceutical manufacturing.
Over the decades, fermentation expanded into:
Vitamin production
Amino acids
Steroids
Hormones
Biologics
Today, Fermentation-Based API manufacturing is a cornerstone of biotechnology-driven pharma plants.
Types of Fermentation Used in API Manufacturing
Fermentation-Based API production relies on carefully selected fermentation strategies depending on the product type, microorganism behavior, yield requirements, and regulatory considerations. The three main fermentation modes used in pharmaceutical manufacturing are batch, fed-batch, and continuous fermentation. Each has distinct operational characteristics, advantages, and industrial applications.
1. Batch Fermentation
In batch fermentation, all required nutrients and microorganisms are added to the bioreactor at the beginning of the process. The system is then closed, and the fermentation proceeds without adding additional nutrients during the run. The microorganisms grow, metabolize the substrate, and produce the desired Active Pharmaceutical Ingredient until the nutrients are depleted or the product concentration reaches its peak.
This method is widely used in Fermentation-Based API manufacturing because it is straightforward to operate and easy to validate under GMP conditions. Since the system remains closed after inoculation, the risk of contamination is relatively low compared to other fermentation types. Batch fermentation is particularly suitable for antibiotic production, including drugs like Penicillin and Streptomycin.
During the batch process, microbial growth typically follows distinct phases:
Lag phase (adaptation)
Log phase (rapid growth)
Stationary phase (maximum product formation)
Decline phase (nutrient exhaustion)
Most antibiotics are secondary metabolites produced during the stationary phase, making batch fermentation ideal for such APIs.
Advantages of Batch Fermentation:
Simple operation and process control
Lower contamination risk
Easier regulatory validation
Suitable for antibiotic production
Flexible for small-to-medium scale manufacturing
However, batch fermentation may result in lower productivity compared to fed-batch or continuous systems because nutrient levels cannot be optimized during the process.
2. Fed-Batch Fermentation
Fed-batch fermentation is an advanced and highly optimized method used extensively in modern Fermentation-Based API facilities. In this process, nutrients are added gradually during fermentation instead of being supplied all at once. This controlled feeding strategy prevents substrate inhibition, maintains optimal microbial growth, and enhances product yield.
By carefully regulating the feed rate, manufacturers can control parameters such as dissolved oxygen demand, metabolic rate, and by-product formation. This leads to higher productivity and improved process stability.
Fed-batch fermentation is widely used in the production of recombinant proteins and hormones such as Insulin. It is especially valuable when producing high-cell-density cultures where nutrient balance is critical for maximum API output.
There are different feeding strategies used in fed-batch systems:
Constant feed rate
Exponential feeding
Feedback-controlled feeding (based on pH or dissolved oxygen)
This flexibility makes fed-batch fermentation the most commonly adopted process in biopharmaceutical manufacturing today.
Advantages of Fed-Batch Fermentation:
Higher product yield
Better control over growth conditions
Reduced substrate inhibition
Suitable for recombinant and high-value APIs
Improved scalability
Although more complex than batch systems, fed-batch fermentation provides superior productivity and is often preferred for large-scale commercial API manufacturing.
3. Continuous Fermentation
Continuous fermentation is a process in which fresh sterile media is continuously added to the bioreactor while an equal volume of culture broth is simultaneously removed. This maintains the culture at a steady state, where microbial growth and product formation occur continuously over extended periods.
In continuous Fermentation-Based API systems, parameters such as dilution rate, nutrient concentration, and oxygen supply must be precisely controlled. The system operates at equilibrium, ensuring consistent production output. This method is particularly beneficial for APIs that are growth-associated products.
Continuous fermentation offers very high productivity and is efficient for large-scale operations where consistent demand exists. However, maintaining sterility over long operational periods can be challenging, and contamination risks are higher compared to batch processes.
This method is gaining attention in modern bioprocessing as pharmaceutical manufacturers move toward continuous manufacturing to improve efficiency and reduce production costs.
Advantages of Continuous Fermentation:
High productivity and output
Efficient for large-scale manufacturing
Reduced downtime between batches
Consistent product quality (under stable conditions)
Challenges:
Complex process control
Higher contamination risk
Difficult validation under strict regulatory standards
Step-by-Step Fermentation-Based API Manufacturing Process
Below is a detailed explanation of each stage involved in Fermentation-Based API production. Each step plays a critical role in ensuring high yield, purity, and regulatory compliance.
1. Strain Selection and Development
High-producing microbial strain selected
Genetic modification (if required)
Stability and productivity validation
2. Media Preparation
Carbon source (glucose, starch)
Nitrogen source (ammonium salts)
Trace elements and minerals
Sterilization via autoclave or SIP
The fermentation media provides essential nutrients required for microbial growth and product formation. The carbon source supplies energy, while nitrogen supports protein synthesis and cell development. Trace elements such as magnesium, iron, and zinc are added to support enzymatic functions. The prepared media must be sterilized using autoclaves or Steam-In-Place (SIP) systems to eliminate contamination before entering the bioreactor.
3. Inoculum Development
Small-scale seed culture
Scale-up in seed fermenters
Transfer to production bioreactor
Inoculum development ensures that an active and healthy microbial population is introduced into the production fermenter. The process begins with a small laboratory seed culture, which is gradually scaled up in seed fermenters to increase biomass concentration. This step is critical to achieving optimal fermentation performance and reducing lag time in large-scale production.
4. Fermentation Stage
Critical parameters controlled:
Temperature
pH
Dissolved oxygen
Agitation speed
Aeration rate
Typical fermentation time: 2–10 days.
During the fermentation stage, microorganisms convert nutrients into the desired Active Pharmaceutical Ingredient. Precise control of process parameters is essential to maintain optimal microbial activity and maximize yield. Advanced bioreactors equipped with sensors and automated control systems continuously monitor conditions to ensure stable production throughout the fermentation cycle.
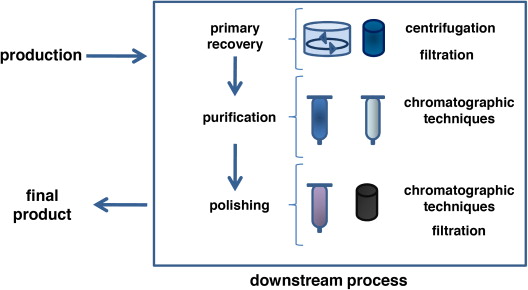
5. Harvesting
Broth separation via centrifugation
Filtration of biomass
After fermentation is complete, the culture broth contains the target API along with cells and residual media components. Harvesting involves separating microbial biomass from the liquid phase using centrifuges or filtration systems. Efficient separation improves downstream purification efficiency and reduces processing time.
6. Downstream Processing
Includes:
Extraction
Purification
Crystallization
Drying
Downstream processing is one of the most critical and cost-intensive stages of Fermentation-Based API manufacturing. The target compound is extracted and purified to remove impurities, residual solvents, and unwanted by-products. Crystallization enhances product purity and consistency, while drying ensures stability and proper storage conditions.
7. Final API Formulation
The purified API is milled, blended, and standardized to meet pharmacopeial specifications. Final quality control tests are performed to verify potency, purity, moisture content, and compliance with regulatory standards before packaging and release for formulation into finished dosage forms.
Equipment Used in Fermentation-Based API Plants
Fermentation-Based API manufacturing requires highly specialized and GMP-compliant equipment to ensure sterile processing, consistent yield, and regulatory compliance. Since the process involves live microorganisms and sensitive biological reactions, equipment selection plays a critical role in productivity, contamination control, and product quality.
Major equipment includes:
Bioreactors (SS316L)
Seed fermenters
Centrifuges
Filtration systems
Spray dryers
Lyophilizers
CIP/SIP systems
Cleanroom facilities
Plant design must follow GMP standards.
Quality Control in Fermentation-Based API
Quality Control in Fermentation-Based API manufacturing involves rigorous testing and monitoring at multiple stages to ensure consistent product quality and safety. During fermentation, in-process controls such as pH, temperature, dissolved oxygen, microbial count, and metabolite concentration are continuously monitored. Raw materials are tested for identity, purity, and microbial contamination before use. After production, the final API undergoes comprehensive analytical testing, including assay determination, impurity profiling, residual solvent analysis, endotoxin testing, moisture content evaluation, and microbiological examination. These tests are performed according to pharmacopeial standards (such as IP, USP, EP) and validated analytical methods to ensure compliance with global regulatory requirements.
Tests include:
Microbial purity
Potency assay
Residual solvent analysis
Endotoxin testing
Moisture content
Stability studies
Regulatory authorities require strict documentation.
Regulatory Requirements
Fermentation-Based API manufacturing must comply with:
US Food and Drug Administration (FDA)
European Medicines Agency (EMA)
Central Drugs Standard Control Organization (CDSCO)
Key documentation:
DMF (Drug Master File)
Process validation report
Batch manufacturing record
Environmental monitoring logs
Change control documentation
Advantages of Fermentation-Based API
Environmentally friendly
Lower energy consumption
High selectivity
Reduced hazardous waste
Sustainable raw materials
Fermentation supports green chemistry principles.
Challenges in Fermentation-Based API Manufacturing
Contamination risk
High capital cost
Complex scale-up
Batch variability
Strict regulatory scrutiny
Process optimization and automation reduce risks.
Applications of Fermentation-Based API
1. Antibiotics
Penicillin
Cephalosporin
2. Hormones
Insulin
3. Vaccines
Hepatitis B vaccine
4. Enzymes
Proteases
Amylases
5. Biologics
Monoclonal antibodies
Design Considerations for Fermentation API Plants
Since you frequently write about pharmaceutical plant design, here are critical considerations:
Facility Layout
Unidirectional flow
Segregation of clean/dirty areas
HVAC zoning
Utilities
Purified water
WFI
Clean steam
Compressed air
Containment
Biosafety level compliance
Closed systems
Automation
SCADA systems
PLC control
Environmental and Waste Management
Fermentation generates:
Spent biomass
Wastewater
CO₂
Treatment methods include:
Effluent treatment plants
Biomass composting
Anaerobic digestion
Future Trends in Fermentation-Based API
Synthetic biology
CRISPR gene editing
Continuous bioprocessing
AI-driven fermentation optimization
Single-use bioreactors
Biotechnology is shaping the next generation of API production.
Conclusion
Fermentation-Based API manufacturing is a sustainable, efficient, and technologically advanced method of producing pharmaceutical ingredients. From antibiotics to biologics, fermentation continues to play a vital role in global healthcare.
With strict regulatory compliance, advanced bioreactor systems, and optimized downstream processing, this method ensures high-quality API production with reduced environmental impact.
As the industry shifts toward biotechnology and green manufacturing, fermentation-based processes will become even more critical in the pharmaceutical landscape.
Frequently Asked Questions (FAQ)
1. What is Fermentation-Based API?
Fermentation-Based API refers to pharmaceutical ingredients produced using microbial fermentation processes instead of chemical synthesis.
2. Which microorganisms are used in Fermentation-Based API manufacturing?
Bacteria, yeast, and fungi are commonly used.
3. What are the advantages of Fermentation-Based API?
It offers high selectivity, sustainability, reduced waste, and environmentally friendly production.
4. What is the difference between batch and fed-batch fermentation?
Batch fermentation uses fixed nutrients, while fed-batch adds nutrients during the process to increase yield.
5. Is Fermentation-Based API GMP regulated?
Yes, it must comply with global regulatory authorities like FDA, EMA, and CDSCO.
6. What equipment is required for Fermentation-Based API production?
Bioreactors, centrifuges, filtration systems, dryers, CIP/SIP systems, and cleanroom facilities are required.
7. What challenges exist in Fermentation-Based API manufacturing?
Contamination, scale-up complexity, and regulatory compliance are key challenges.