The API Manufacturing Process is the foundation of modern pharmaceutical production and plays a decisive role in ensuring that medicines are safe, effective, and consistent in quality. API stands for Active Pharmaceutical Ingredient, which is the core substance in any drug responsible for producing its intended therapeutic effect inside the human body. Whether it is a simple pain reliever, a life-saving antibiotic, or a complex anticancer medication, the API is the primary component that delivers the desired biological action.
Without a well-structured and strictly controlled API manufacturing process, pharmaceutical products cannot meet global safety standards, regulatory requirements, or patient expectations. Even minor deviations in temperature, raw material quality, or purification techniques can significantly impact drug performance. Therefore, understanding how APIs are manufactured is essential for students, pharmaceutical professionals, quality assurance teams, and industry stakeholders.

What is API in Pharmaceuticals?
An Active Pharmaceutical Ingredient (API) is the biologically active component of a medicine that directly produces the therapeutic effect. While tablets, capsules, injections, and syrups contain multiple ingredients, only the API is responsible for treating or preventing disease. The remaining components are known as excipients, which help in drug delivery, stability, and absorption but do not have therapeutic effects.
For example:
In a painkiller tablet, the API works to reduce inflammation and relieve pain.
In an antibiotic medication, the API destroys or inhibits the growth of bacteria.
In an antihypertensive drug, the API lowers blood pressure by acting on specific receptors or enzymes.
The API manufacturing process ensures that this active compound is produced with high purity, consistent strength, and minimal impurities. This consistency is critical because variations in API quality can directly affect patient safety and drug effectiveness.
Overview of the API Manufacturing Process
The API manufacturing process typically involves multiple carefully monitored stages. Although the specific methods may vary depending on the drug type, the general steps include:
Raw Material Selection
Chemical Synthesis or Fermentation
Purification
Crystallization
Drying and Milling
Quality Control Testing
Packaging and Storage
Each stage plays a crucial role in ensuring that the final API meets international regulatory standards and pharmacopeial requirements.
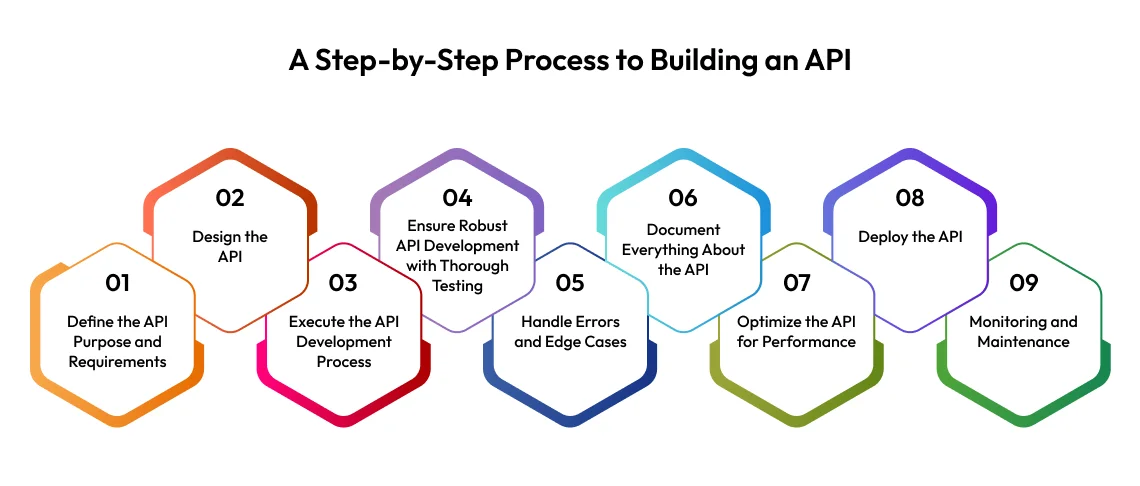
Step-by-Step Process of API Manufacturing
In addition to the technical manufacturing stages, the API manufacturing process also follows a structured development lifecycle. Below is a simplified step-by-step framework that aligns with industry best practices:
1. Define the API Purpose and Requirements
Before manufacturing begins, the therapeutic objective is clearly defined. This includes target disease, dosage strength, safety profile, and regulatory expectations.
2. Design the API
Scientists design the molecular structure and determine the most efficient synthesis or biological pathway for production.
3. Execute the API Development Process
Laboratory-scale synthesis is performed, followed by pilot-scale studies to validate feasibility, scalability, and cost-effectiveness.
4. Ensure Robust API Development with Thorough Testing
Extensive testing is conducted to confirm purity, stability, potency, and impurity profiles. Analytical methods are validated at this stage.
5. Handle Errors and Edge Cases
Risk assessment and process optimization are performed to manage deviations, contamination risks, and unexpected reaction outcomes.
6. Document Everything About the API
Complete documentation is prepared, including batch records, validation reports, stability data, and Drug Master File (DMF) submissions.
7. Optimize the API for Performance
Process parameters are refined to improve yield, reduce waste, enhance efficiency, and ensure consistent quality.
8. Deploy the API
Commercial-scale manufacturing begins after regulatory approval and validation.
9. Monitoring and Maintenance
Ongoing quality monitoring, audits,
stability studies, and regulatory compliance ensure long-term reliability of the API manufacturing process.
1. Raw Material Selection
The API manufacturing process begins with selecting high-quality raw materials, intermediates, reagents, and solvents. The success of the entire production cycle depends heavily on the quality of these starting materials.
Manufacturers carefully evaluate suppliers to ensure reliability and compliance with pharmaceutical standards. Raw materials must meet specific purity levels, and they are tested before use. Documentation such as Certificates of Analysis (CoA) is reviewed to verify compliance with pharmacopeial standards.
Key considerations during this stage include:
Chemical purity and stability of starting materials
Supplier qualification and audit approval
Cost efficiency without compromising quality
Compliance with regulatory guidelines
Poor-quality raw materials can introduce impurities that are difficult to remove later in the process, potentially leading to product rejection or regulatory issues.
2. Chemical Synthesis or Fermentation
This stage is where the actual formation of the API occurs. Depending on the drug type, manufacturers use either chemical synthesis or fermentation.
A. Chemical Synthesis
Chemical synthesis involves carrying out controlled chemical reactions inside industrial reactors. These reactions convert raw materials into the desired API through multiple intermediate stages.
The process includes:
Reaction setup and charging of materials
Temperature and pressure monitoring
Use of catalysts and reagents
Monitoring reaction progress using analytical tools
Isolation of intermediate compounds
This method is commonly used for small-molecule drugs such as analgesics, antihypertensives, and anti-inflammatory medicines. Each reaction step is validated and documented to ensure reproducibility and regulatory compliance.
B. Fermentation
Certain APIs, particularly antibiotics and biologics, are produced using fermentation. In this method, microorganisms such as bacteria or fungi are cultured under controlled conditions to produce the desired compound.
The fermentation process includes:
Preparation of nutrient-rich media
Controlled microbial growth in bioreactors
Regulation of temperature, pH, and oxygen levels
Harvesting the product from the fermentation broth
Fermentation requires strict environmental control to prevent contamination and ensure optimal yield. It is widely used in producing complex biological APIs.
3. Purification Process
After synthesis or fermentation, the crude API contains impurities such as unreacted raw materials, by-products, residual solvents, and degradation compounds. Purification is essential to achieve the required level of purity.
Common purification techniques include:
Filtration to remove solid impurities
Distillation to separate solvents
Chromatography for fine separation
Recrystallization to enhance purity
Impurity control is a critical regulatory requirement. Regulatory authorities set strict limits for specific impurities, especially those that may be toxic or carcinogenic.
4. Crystallization
Crystallization is an important step that improves both purity and physical properties of the API. During this stage, the purified compound is converted into a crystalline solid form.
This process helps in:
Controlling particle size
Ensuring consistent crystal structure
Improving solubility and stability
Enhancing downstream processing
Different crystal forms, known as polymorphs, can influence drug performance. Therefore, crystallization conditions are carefully optimized and controlled.
5. Drying and Milling
Once crystallized, the API must be dried to remove residual solvents and moisture. Excess moisture can cause degradation or affect shelf life.
After drying, milling is performed to achieve the desired particle size distribution. Particle size plays a crucial role in drug formulation because it influences dissolution rate, bioavailability, and absorption in the body.
Modern equipment ensures uniform particle size and minimizes contamination risks during processing.
6. Quality Control (QC) Testing
Quality Control is one of the most critical stages in the API manufacturing process. Every batch undergoes rigorous testing before release.
Common tests include:
Assay testing to measure API strength
Impurity profiling
Residual solvent analysis
Heavy metal testing
Microbial testing (if required)
Analytical techniques used include:
HPLC (High-Performance Liquid Chromatography)
GC (Gas Chromatography)
UV Spectroscopy
IR Spectroscopy
APIs must comply with pharmacopeial standards such as the United States Pharmacopeia (USP), European Medicines Agency (EMA guidelines), and Indian Pharmacopeia requirements. Any deviation can result in batch rejection.
7. Packaging and Storage
After passing Quality Assurance approval, the API is packed in suitable containers that protect it from moisture, light, and contamination. Packaging materials are selected based on compatibility and stability data.
Proper labeling includes batch number, manufacturing date, expiry date, storage conditions, and regulatory details. APIs are stored in controlled environments with specific temperature and humidity conditions to maintain stability throughout their shelf life.
Types of API Manufacturing
1. Synthetic APIs
Produced entirely through chemical reactions.
2. Semi-Synthetic APIs
Derived from natural sources and chemically modified.
3. Biotech APIs
Produced using biological systems such as cells, enzymes, or microorganisms.
Each type requires different infrastructure, expertise, and regulatory controls.
Regulatory Requirements in API Manufacturing
The API manufacturing process must comply with strict global regulations, including:
ICH Guidelines
Drug Master File (DMF) submission
Stability studies
Regulatory bodies such as the U.S. Food and Drug Administration (FDA), European Medicines Agency (EMA), and Central Drugs Standard Control Organization (CDSCO) monitor compliance.
Failure to meet these standards can result in warning letters, import bans, product recalls, or facility shutdown.
Challenges in API Manufacturing
The pharmaceutical industry faces several challenges in API production, including:
Maintaining consistent batch quality
Controlling impurities within permissible limits
Managing supply chain disruptions
Ensuring environmental compliance
Handling high-potency compounds safely
To address these challenges, manufacturers increasingly adopt automation, real-time monitoring systems, and digital quality management platforms.
Importance of API Manufacturing in the Pharmaceutical Industry
The API manufacturing process ensures drug safety, therapeutic effectiveness, and global regulatory acceptance. Since you are based in India, it is important to note that India is one of the largest API manufacturing production hubs globally, supplying APIs to major pharmaceutical markets worldwide. Maintaining international quality standards is essential for export approvals and global competitiveness.
A minor deviation in synthesis conditions or impurity control can significantly affect drug efficacy and patient health. Therefore, robust process design and quality assurance are essential components of successful API production.
Future Trends in API Manufacturing
The future of API manufacturing is focused on innovation and sustainability. Key emerging trends include:
Continuous manufacturing processes
Green chemistry approaches to reduce waste
Automation and AI-based monitoring systems
High-potency API (HPAPI) facilities
Environmentally sustainable production methods
These advancements aim to improve efficiency, reduce environmental impact, and enhance product consistency.
Conclusion
The API Manufacturing Process is a complex, multi-stage operation that forms the backbone of pharmaceutical production. From selecting raw materials to final packaging, every step must follow strict quality and regulatory standards. Ensuring purity, consistency, and compliance is not only a legal requirement but also a responsibility toward patient safety.
By understanding the API manufacturing process in depth, pharmaceutical students, professionals, and business owners can better appreciate how medicines are developed, tested, and safely delivered to patients around the world.
1. What is an API in pharmaceuticals?
An API (Active Pharmaceutical Ingredient) is the main component in a medicine that produces the intended therapeutic effect. It is the substance that treats, cures, prevents, or diagnoses a disease. Without the API, a medicine would not have any medical benefit.
2. What is the API manufacturing process?
The API manufacturing process is the step-by-step procedure used to produce the active ingredient of a drug. It includes raw material selection, synthesis or fermentation, purification, crystallization, drying, milling, quality control testing, and packaging. Each step is strictly controlled to ensure safety, purity, and consistency.
3. What is the difference between API and excipients?
API is the active substance that produces the therapeutic effect, while excipients are inactive ingredients that help deliver the drug effectively. Excipients improve stability, absorption, taste, and shelf life but do not treat the disease.
4. What are the main types of API manufacturing?
There are three main types:
Synthetic APIs – Produced through chemical reactions.
Semi-synthetic APIs – Derived from natural sources and chemically modified.
Biotech APIs – Produced using biological systems such as microorganisms or cell cultures.
5. What is the difference between chemical synthesis and fermentation?
Chemical synthesis involves controlled chemical reactions in reactors to form the API. It is commonly used for small-molecule drugs.
Fermentation uses microorganisms to produce APIs under controlled environmental conditions. It is often used for antibiotics and biologics.
6. Why is purification important in API manufacturing?
Purification removes impurities such as unreacted materials, by-products, and residual solvents. Regulatory authorities set strict limits for impurities because they can affect drug safety and effectiveness. High purity ensures patient safety and compliance with global standards.
7. What is GMP in API manufacturing?
GMP stands for Good Manufacturing Practices. It is a system that ensures products are consistently produced and controlled according to quality standards. Regulatory bodies like the U.S. Food and Drug Administration (FDA) and the Central Drugs Standard Control Organization (CDSCO) require manufacturers to follow GMP guidelines.