Critical Process Parameters (CPP) are the backbone of any controlled and consistent manufacturing process. If you’re aiming to produce high-quality products without variation, understanding Critical Process Parameters (CPP) isn’t optional—it’s essential. These parameters represent the key variables that directly influence the outcome of a process, and if they drift outside defined limits, the final product can fail to meet quality standards.
Think of Critical Process Parameters (CPP) like the controls on a complex machine. Every dial, every setting, and every adjustment plays a role in determining the final result. Whether it’s temperature, pressure, mixing speed, or time, each of these variables must be carefully managed to ensure consistency. Even a slight variation in one of these Critical Process Parameters (CPP) can lead to defects, inefficiencies, or compliance issues.
In industries like pharmaceuticals and food production, Critical Process Parameters (CPP) are tightly regulated. Authorities such as the FDA require companies to clearly define, monitor, and control these parameters to ensure safety and effectiveness. This makes CPP not just a technical concept, but a regulatory necessity.
Here’s why Critical Process Parameters (CPP) are so important:
- They directly impact product quality and safety
- They ensure consistency across production batches
- They help meet regulatory compliance standards
- They reduce waste and improve efficiency
Understanding Critical Process Parameters (CPP) also helps businesses move from reactive problem-solving to proactive process control. Instead of fixing issues after they occur, companies can prevent them by maintaining tight control over these critical variables.

Why CPP Matters in Modern Industries
Critical Process Parameters (CPP) play a central role in ensuring that manufacturing processes remain efficient, predictable, and scalable. Businesses that prioritize Critical Process Parameters (CPP) are better equipped to handle variability, reduce waste, and maintain consistent output quality. By continuously refining Critical Process Parameters (CPP), organizations can adapt to changing production demands without compromising performance.
Another major factor is compliance. Regulatory agencies require detailed documentation and control of Critical Process Parameters to ensure public safety. Failing to meet these requirements can result in fines, product recalls, or even shutdowns. In industries like pharmaceuticals, this isn’t just about business—it’s about protecting lives.
There’s also a financial angle to consider. Poor control of Critical Process Parameters (CPP) can lead to waste, rework, and inefficiencies. Imagine producing an entire batch of products only to discover they don’t meet quality standards due to a minor parameter deviation. That’s time, money, and resources down the drain.
On the flip side, well-managed CPPs can significantly improve operational efficiency. Companies can optimize processes, reduce downtime, and even predict potential issues before they occur. With advancements in automation and real-time monitoring, managing Critical Process Parameters (CPP) has become more precise than ever.
In short, CPPs are the backbone of modern production systems. They bridge the gap between process design and product quality, ensuring that everything runs smoothly from start to finish.
Understanding the Role of CPP in Process Control
CPP vs Critical Quality Attributes (CQA)
If you’ve been exploring Critical Process Parameters (CPP) for a while, you’ve probably come across another closely related term: Critical Quality Attributes (CQA). At first glance, they might seem interchangeable, but they serve very different roles in process management. Understanding the relationship between these two is essential if you want to truly master process optimization.
Think of Critical Quality Attributes as the “end goal” and Critical Process Parameters (CPP) as the “means to get there.” CQAs define the physical, chemical, or biological properties that a product must have to meet safety and performance standards. These could include things like purity, strength, or texture. On the other hand, Critical Process Parameters (CPP) are the variables you control during production to ensure those attributes are consistently achieved.
For example, in pharmaceutical manufacturing, the dissolution rate of a tablet might be a CQA. To achieve the desired dissolution rate, you would carefully control Critical Process Parameters (CPP) such as compression force, granulation moisture content, and drying temperature. If any of these CPPs go out of range, the final product might fail to meet its quality requirements.
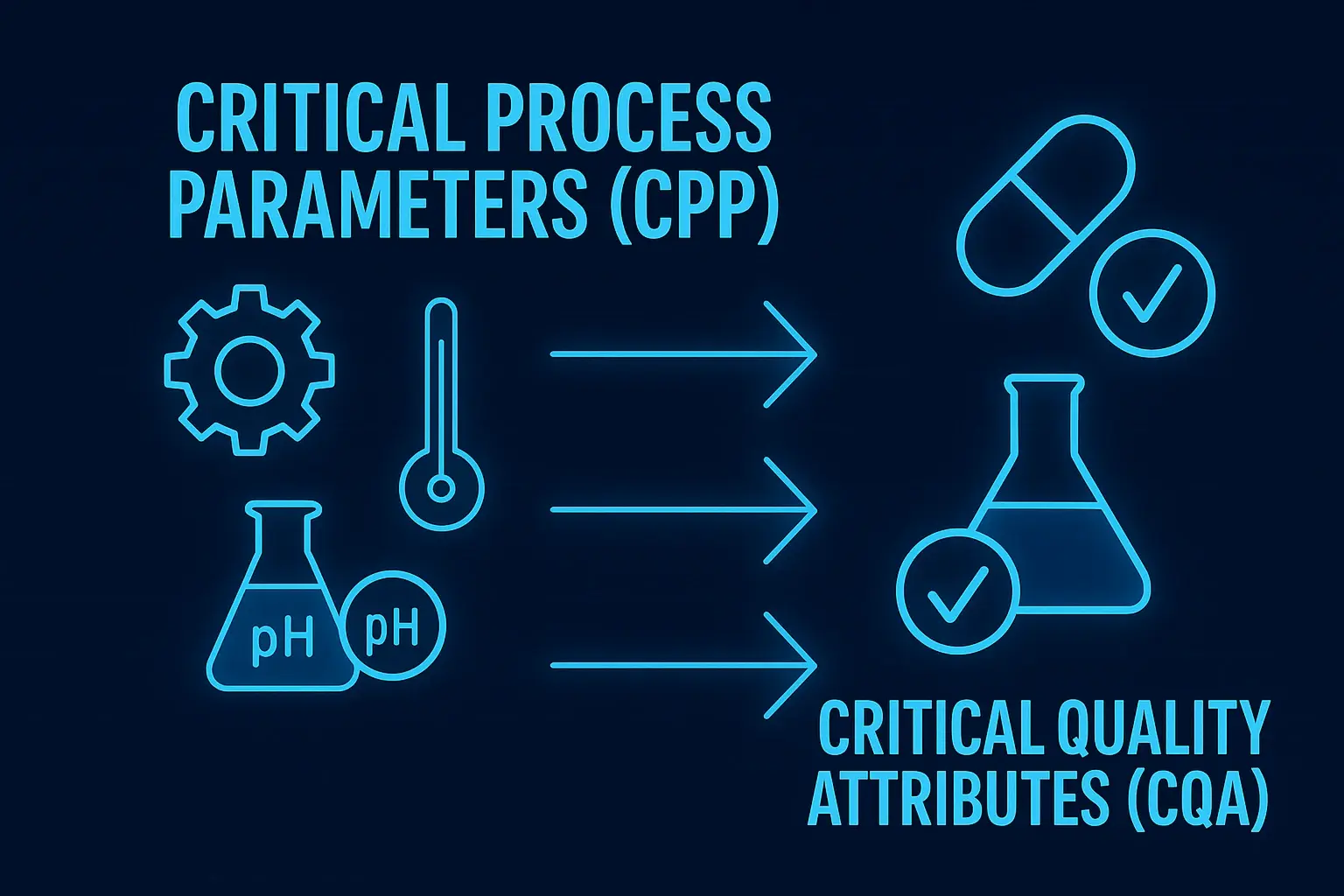
Here’s a simple breakdown to clarify the distinction:
| Aspect | Critical Process Parameters (CPP) | Critical Quality Attributes (CQA) |
|---|---|---|
| Definition | Process variables that impact quality | Product characteristics that define quality |
| Focus | During manufacturing | Final product outcome |
| Examples | Temperature, pressure, mixing speed | Purity, potency, appearance |
| Role | Control and optimization | Quality evaluation |
Understanding this relationship allows companies to design more efficient processes. By identifying which Critical Process Parameters directly influence CQAs, organizations can focus their efforts on controlling what truly matters, rather than wasting resources on less impactful variables.
- Key takeaway: CPPs drive CQAs, and controlling CPPs ensures consistent product quality.
- Practical tip: Always map each CQA to one or more Critical Process Parameters during process design.
- Industry insight: Regulatory guidelines like ICH Q8 emphasize the link between CPP and CQA.
When you connect these dots, process control becomes much more strategic rather than reactive.
The Science Behind Process Stability
Process stability isn’t just a buzzword—it’s the backbone of consistent production, and it relies heavily on how well you manage Critical Process Parameters. Without stability, even the most advanced systems can produce inconsistent or defective products. So what exactly makes a process stable?
At its core, stability means that a process performs predictably over time. This predictability comes from maintaining Critical Process Parameters within defined limits. When these parameters fluctuate too much, variability increases, and that’s when problems start to appear.
Imagine trying to bake bread in an oven where the temperature randomly spikes or drops. Even if you follow the same recipe, the results will vary. The same principle applies in industrial processes—if Critical Process Parameters like temperature or pressure aren’t tightly controlled, the output becomes unreliable.
Several scientific principles support process stability:
- Statistical Process Control (SPC): Uses data to monitor and control CPP variations.
- Process Capability Analysis: Measures how well a process can meet specifications.
- Control Charts: Help visualize trends and detect deviations early.
Modern industries rely heavily on these tools to maintain control over Critical Process Parameters. By continuously monitoring data, companies can detect even the smallest shifts and take corrective action before issues escalate.
Here’s why process stability matters so much:
- It reduces product variability and defects
- It improves efficiency and minimizes waste
- It ensures compliance with regulatory standards
- It builds customer trust through consistent quality
Another critical aspect is the concept of a design space. This is the range within which Critical Process Parameters can vary without affecting product quality. Operating within this space ensures flexibility while maintaining control.
Ultimately, mastering the science behind process stability means understanding how each parameter interacts with the others. It’s not just about controlling individual variables—it’s about managing the entire system as a cohesive unit.
Key Components of Critical Process Parameters
Temperature, Pressure, and Flow Rate
When discussing Critical Process Parameters (CPP) , certain variables come up again and again—and for good reason. Temperature, pressure, and flow rate are among the most influential factors in many industrial processes. These parameters often serve as the foundation for maintaining control and ensuring product consistency.
Let’s start with temperature. It’s one of the most sensitive Critical Process Parameters (CPP) because even slight deviations can have a significant impact. In chemical reactions, temperature affects reaction rates, product yield optimization, and even safety. Too high, and you risk degradation or hazardous conditions; too low, and the process may become inefficient or incomplete.
Pressure is another key player. In processes like distillation or filtration, pressure directly influences how materials behave. Maintaining the correct pressure ensures that reactions occur under optimal conditions and that equipment operates safely.
Flow rate, meanwhile, determines how materials move through a system. Whether it’s liquids, gases, or powders, controlling flow rate ensures proper mixing, reaction time, and overall efficiency.
Here’s a quick overview:
- Temperature
- Affects reaction speed and product stability
- Requires precise monitoring and control systems
- Pressure
- Impacts phase changes and reaction conditions
- Critical for safety and equipment integrity
- Flow Rate
- Controls material movement and process timing
- Essential for uniformity and consistency
These three parameters often work together. For example, increasing temperature might require adjustments in pressure or flow rate to maintain balance. That’s why managing Critical Process Parameters (CPP) isn’t about isolated control—it’s about understanding their interdependence.
Industries invest heavily in sensors and automation to monitor these variables in real time. Advanced systems can even make automatic adjustments, ensuring that Critical Process Parameters (CPP) remain within acceptable limits without manual intervention.
Time, pH, and Environmental Factors
While temperature and pressure often get the spotlight, other Critical Process Parameters like time, pH, and environmental conditions are just as important. These variables might seem secondary, but they can have a profound impact on product quality and process efficiency.
Time is a deceptively simple parameter. Whether it’s reaction time, mixing duration, or drying period, timing plays a crucial role in determining outcomes. Too short, and the process may be incomplete; too long, and you risk overprocessing or degradation.
pH is especially critical in industries like pharmaceuticals and food production. It affects chemical stability, microbial growth, and overall product safety. Maintaining the correct pH level ensures that the product remains effective and safe for consumption.
Environmental factors—such as humidity, contamination levels, and air quality—also fall under Critical Process Parameters in many cases. These conditions can influence everything from material stability to equipment performance.
Key considerations include:
- Time
- Determines process completion and efficiency
- Must be optimized for each stage
- pH Levels
- Influences chemical reactions and stability
- Requires continuous manufacturing
- Environmental Conditions
- Includes humidity, cleanliness, and temperature control
- Critical in controlled environments like cleanrooms
One important thing to remember is that these parameters often interact. For example, changes in humidity can affect drying time, which in turn impacts product quality. Managing Critical Process Parameters effectively means understanding these relationships and adjusting accordingly.
Companies that overlook these “secondary” parameters often face unexpected issues. That’s why a comprehensive approach to CPP management is essential—every variable matters.
How to Identify Critical Process Parameters
Risk Assessment Methods
Identifying Critical Process Parameters (CPP) isn’t something you guess—it’s a structured, data-driven process that requires careful evaluation of risks. If you skip this step or rush through it, you’re essentially building your entire process control strategy on shaky ground. The goal here is simple: figure out which parameters have the highest impact on product quality and prioritize them.
Risk assessment methods are the backbone of this identification process. They help you systematically analyze every variable involved in production and determine which ones truly matter. One of the most widely used approaches is Failure Mode and Effects Analysis (FMEA). This method evaluates potential failure points, their causes, and their impact on the final product. By assigning risk scores, teams can easily identify which Critical Process Parameters (CPP) need strict monitoring.
Another powerful tool is Hazard Analysis and Critical Control Points (HACCP), especially popular in food and pharmaceutical industries. HACCP focuses on identifying hazards and implementing controls at critical stages of the process. This makes it highly effective for pinpointing Critical Process Parameters (CPP) that directly affect safety and compliance.
Here are some commonly used risk assessment techniques:
- FMEA (Failure Mode and Effects Analysis)
Helps prioritize risks based on severity, occurrence, and detection - HACCP (Hazard Analysis Critical Control Points)
Focuses on safety-critical parameters - Risk Ranking and Filtering
Simplifies complex processes by ranking variables - Fault Tree Analysis (FTA)
Identifies root causes of failures
The real advantage of these methods is clarity. Instead of treating all process variables equally, you can focus on the few that truly drive outcomes. This not only improves efficiency but also reduces monitoring costs.
Another important aspect is cross-functional collaboration. Engineers, quality experts, and operators should all be involved in identifying Critical Process Parameters (CPP) . Each team brings a different perspective, and together they can uncover risks that might otherwise be overlooked.
Ultimately, risk assessment transforms uncertainty into actionable insights. It gives you a clear roadmap for controlling your process and ensures that your Critical Process Parameters (CPP) are backed by solid reasoning—not assumptions.
Design of Experiments (DoE) Approach
While risk assessment helps you narrow down potential Critical Process Parameters (CPP) , the Design of Experiments (DoE) approach takes things a step further by validating those assumptions with data. If risk assessment is about identifying possibilities, DoE is about proving what actually works.
DoE is a structured method for testing multiple variables simultaneously. Instead of changing one factor at a time, you experiment with combinations of variables to see how they interact and affect the outcome. This is especially useful when dealing with complex processes where Critical Process Parameters (CPP) don’t operate in isolation.
For example, let’s say you’re trying to optimize a mixing process. Instead of testing temperature, speed, and time separately, DoE allows you to analyze how all three factors interact. You might discover that a slight increase in temperature only works if the mixing speed is also adjusted. These insights are invaluable for defining accurate Critical Process Parameters (CPP).
Key benefits of using DoE include:
- Efficient experimentation
Reduces the number of trials needed - Interaction analysis
Identifies relationships between variables - Data-driven decisions
Eliminates guesswork - Process optimization
Helps define ideal operating conditions
There are different types of DoE methods, such as:
- Full factorial designs
- Fractional factorial designs
- Response surface methodology
Each method serves a specific purpose depending on the complexity of the process.
What makes DoE particularly powerful is its ability to define a design space—the range within which Critical Process Parameters (CPP) can vary without affecting product quality. This gives manufacturers flexibility while maintaining control.
Companies that rely on DoE often see significant improvements in efficiency and consistency. Instead of constantly reacting to problems, they proactively design processes that are robust and reliable.
CPP in Different Industries
Pharmaceutical Industry Applications
The pharmaceutical industry is perhaps the most rigorous when it comes to managing Critical Process Parameters (CPP). And for good reason—lives depend on it. Every tablet, injection, or capsule must meet strict quality standards, and even the smallest deviation in a parameter can lead to serious consequences.
In pharma, Critical Process Parameters are identified during the drug development phase and are closely linked to Critical Quality Attributes (CQA). Regulatory guidelines such as ICH Q8, Q9, and Q10 emphasize the importance of defining and controlling these parameters throughout the product lifecycle.
For example, in tablet manufacturing, several Critical Process Parameters must be controlled:
- Granulation moisture content
- Drying temperature
- Compression force
- Coating thickness
Each of these parameters directly affects the final product’s quality, including its stability, dissolution rate, and bioavailability.
Pharmaceutical companies also rely heavily on Process Analytical Technology (PAT). This involves real-time monitoring of Critical Process Parameters to ensure they remain within acceptable limits. If a deviation occurs, corrective actions can be taken immediately, reducing the risk of batch failure.
Another key aspect is validation. Before a process is approved, it must demonstrate that it can consistently produce products meeting specifications. This involves extensive testing and documentation of Critical Process Parameters (CPP) .
Here’s why CPP management is so critical in pharma:
- Ensures patient safety
- Meets strict regulatory requirements
- Reduces batch failures and recalls
- Supports continuous improvement
In this industry, there’s no room for error. That’s why Critical Process Parameters (CPP) are treated with the highest level of importance and precision.
Manufacturing and Food Industry Use Cases
While pharmaceuticals often get the spotlight, Critical Process Parameters play an equally important role in manufacturing and food production. These industries may not always face the same level of regulation, but the need for consistency, efficiency, and safety is just as critical.
In manufacturing, Critical Process Parameters can include variables like machine speed, cutting temperature, and material feed rate. These parameters directly influence product dimensions, strength, and finish. For example, in automotive manufacturing, even a slight variation in welding temperature can compromise structural integrity.
In the food industry, CPPs are closely tied to safety and quality. Parameters such as cooking temperature, storage conditions, and pH levels must be carefully controlled to prevent contamination and ensure product freshness.
Common Critical Process Parameters (CPP) in these industries include:
- Manufacturing
- Machine calibration
- Production speed
- Material consistency
- Food Industry
- Cooking and storage temperature
- Moisture levels
- Hygiene conditions
One interesting trend is the increasing use of automation. Smart sensors and IoT devices now allow companies to monitor Critical Process Parameters in real time. This not only improves accuracy but also reduces human error.
Another benefit is scalability. Once Critical Process Parameters (CPP) are well-defined, companies can scale up studies production without compromising quality. This is especially important for growing businesses.
In both manufacturing and food industries, the goal is the same: deliver consistent, high-quality products while minimizing waste and costs. And that’s exactly what effective CPP management makes possible.
Monitoring and Controlling Critical Process Parameters
Tools and Technologies for CPP Monitoring
Keeping Critical Process Parameters (CPP) under control isn’t just about knowing what they are—it’s about having the right tools to track and manage them continuously. Without proper monitoring systems, even well-defined Critical Process Parameters can drift out of range without anyone noticing until it’s too late. That’s where modern tools and technologies step in, transforming process control from reactive troubleshooting into proactive management.
Today’s industries rely heavily on advanced instrumentation and digital systems to monitor Critical Process Parameters in real time. Sensors are the foundation of this setup. These devices measure variables like temperature, pressure, flow rate, and pH with high precision. The data collected is then transmitted to centralized systems where it can be analyzed and visualized.
One of the most widely used systems is SCADA (Supervisory Control and Data Acquisition). SCADA platforms allow operators to monitor multiple Critical Process Parameters from a single interface. They provide real-time alerts, historical data tracking, and trend analysis, making it easier to detect anomalies before they escalate.
Another essential technology is Distributed Control Systems (DCS). Unlike SCADA, which is often used for broader monitoring, DCS is designed for continuous, automated control of processes. It ensures that Critical Process Parameters remain within predefined limits by making automatic adjustments whenever necessary.
Here are some key tools used for monitoring CPP:
- Sensors and Probes
Measure real-time values of process variables - SCADA Systems
Provide centralized monitoring and data visualization - DCS (Distributed Control Systems)
Enable automated control and stability - Data Historians
Store long-term data for analysis and compliance - Process Analytical Technology (PAT)
Common in pharma for real-time quality assurance
What makes these tools powerful is their ability to integrate with each other. For instance, sensor data can feed into a SCADA system, which then communicates with a DCS to make adjustments. This interconnected ecosystem ensures that Critical Process Parameters are always within control.
Another major advantage is predictive capability. With enough data, companies can use analytics to forecast potential deviations in Critical Process Parameters and take preventive action. This reduces downtime and improves efficiency significantly.
In short, the right tools don’t just monitor CPP—they elevate the entire process to a smarter, more reliable level.
Automation and Real-Time Data Tracking
Automation has completely changed how industries manage Critical Process Parameters (CPP). What once required constant manual oversight can now be handled by intelligent systems that monitor, analyze, and adjust parameters in real time. This shift has not only improved efficiency but also significantly reduced human error.
Real-time data tracking is at the heart of this transformation. Instead of relying on periodic checks, modern systems continuously collect data on Critical Process Parameters and update dashboards instantly. This allows operators to see exactly what’s happening at any given moment.
Imagine running a production line where temperature suddenly spikes. In a manual system, this might go unnoticed until a batch is compromised. In an automated setup, the system detects the deviation immediately and either alerts the operator or corrects it automatically. That’s the power of real-time control.
Automation in CPP management typically includes:
- Automated feedback loops
Adjust parameters instantly based on sensor data - Alarm systems
Notify operators when limits are exceeded - Machine learning algorithms
Predict trends and optimize performance - Remote monitoring
Access data from anywhere via cloud platforms
One of the most exciting developments is the integration of Artificial Intelligence (AI). AI can analyze massive datasets to identify patterns that humans might miss. For example, it can detect subtle correlations between different Critical Process Parameters and suggest optimizations.
Here’s why automation is becoming essential:
- Reduces manual intervention
- Improves accuracy and consistency
- Enhances response time to deviations
- Supports scalability and growth
However, automation doesn’t eliminate the need for human expertise. Instead, it enhances it. Engineers and operators can focus on strategic decisions while the system handles routine monitoring of Critical Process Parameters.
The result? A more efficient, reliable, and future-ready process control system.
Challenges in Managing Critical Process Parameters
Common Mistakes and Risks
Managing Critical Process Parameters (CPP) sounds straightforward on paper, but in practice, it comes with its fair share of challenges. Many organizations struggle not because they lack technology, but because they overlook fundamental principles. Even small mistakes in handling Critical Process Parameters (CPP) can lead to significant quality issues, compliance failures, and financial losses.
One of the most common mistakes is misidentifying CPPs. Not every process variable is critical, yet some companies either overclassify or underclassify parameters. Overloading systems with too many “critical” variables can dilute focus, while missing key Critical Process Parameters (CPP) can leave major risks unaddressed.
Another frequent issue is inadequate monitoring. Defining Critical Process Parameters (CPP) is only half the job—if they aren’t continuously tracked, their value diminishes. Some organizations rely on outdated or manual systems, which increases the chances of missing deviations.
Here are some common risks associated with poor CPP management:
- Lack of proper documentation
Leads to inconsistencies and compliance issues - Infrequent calibration of instruments
Results in inaccurate data - Ignoring data trends
Misses early warning signs - Poor training of personnel
Causes operational errors - Failure to update CPPs
Processes evolve, but parameters remain outdated
Another overlooked challenge is data overload. Modern systems generate massive amounts of data, and without proper analysis, it becomes overwhelming. Companies may collect data on Critical Process Parameters (CPP) but fail to extract meaningful insights.
There’s also the human factor. Resistance to change, lack of awareness, and inadequate training can all hinder effective CPP management. Even the best systems won’t work if people don’t use them correctly.
Ultimately, managing Critical Process Parameters (CPP) requires a balance of technology, expertise, and discipline. Ignoring any one of these can lead to costly consequences.
How to Overcome CPP Challenges
The good news? Most challenges related to Critical Process Parameters (CPP) are entirely solvable with the right approach. It’s not about reinventing the wheel—it’s about applying best practices consistently and leveraging the tools already available.
The first step is proper identification and validation. Use structured methods like risk assessment and Design of Experiments (DoE) to ensure that your Critical Process Parameters (CPP) are accurately defined. This eliminates guesswork and builds a strong foundation.
Next comes continuous monitoring. Investing in modern tools like SCADA, DCS, and real-time analytics ensures that Critical Process Parameters (CPP) are always under control. Automation can further enhance this by reducing reliance on manual processes.
Here are practical strategies to overcome CPP challenges:
- Standardize processes
Create clear SOPs for managing CPP - Regular training programs
Ensure staff understand the importance of CPP - Routine calibration and maintenance
Keep monitoring systems accurate - Data analytics implementation
Turn raw data into actionable insights - Continuous improvement cycles
Regularly review and update CPPs
Another powerful approach is adopting a Quality by Design (QbD) framework. This methodology emphasizes designing processes with Critical Process Parameters in mind from the beginning, rather than fixing issues later.
Collaboration also plays a key role. Bringing together cross-functional teams ensures that all aspects of Critical Process Parameters are considered—from engineering to quality assurance.
Companies that successfully overcome these challenges often see dramatic improvements in efficiency, compliance, and product quality. Managing CPP isn’t just about avoiding problems—it’s about unlocking the full potential of your process.
Benefits of Optimizing Critical Process Parameters
Improved Product Quality and Consistency
When businesses truly understand and control Critical Process Parameters (CPP), one of the most immediate and noticeable benefits is a significant improvement in product quality and consistency. Think about it—customers expect the same experience every single time they use a product. Whether it’s a tablet, a packaged snack, or a mechanical component, consistency isn’t optional anymore; it’s expected. This is exactly where Critical Process Parameters (CPP)step in as the backbone of reliability.
By maintaining tight control over Critical Process Parameters (CPP), companies can reduce variability in production. Variability is the silent enemy of quality. Even minor fluctuations in parameters like temperature, pressure, or mixing time can lead to noticeable differences in the final product. When CPPs are optimized, these fluctuations are minimized, ensuring that each batch meets the same specifications.
Another important aspect is defect reduction. Poorly controlled Critical Process Parameters (CPP) often result in defects, rework, or even complete batch rejection. By optimizing these parameters, companies can drastically lower defect rates. According to industry insights, organizations that implement strong CPP control strategies can reduce production defects by up to 30–50%, depending on the complexity of the process.
Here’s how CPP optimization improves quality:
- Consistent output across batches
- Reduced product variability
- Higher compliance with specifications
- Improved customer satisfaction
There’s also a psychological angle here. When customers trust that your product will perform the same way every time, brand loyalty naturally increases. That trust is built on the invisible work done behind the scenes—specifically, the careful management of Critical Process Parameters.
In industries like pharmaceuticals, this consistency can literally be a matter of life and death. A slight variation in dosage or composition can have serious consequences. That’s why optimizing Critical Process Parameters isn’t just a technical necessity—it’s a responsibility.
Cost Efficiency and Regulatory Compliance
Optimizing Critical Process Parameters (CPP) doesn’t just improve quality—it also has a direct and powerful impact on the bottom line. Many companies initially see CPP management as a compliance requirement, but in reality, it’s a major driver of cost efficiency and operational excellence.
Let’s start with cost savings. When Critical Process Parameters are not properly controlled, the result is often waste—wasted materials, wasted time, and wasted energy. Imagine running a full production batch only to discard it because one parameter went out of range. That’s a costly mistake. By optimizing CPPs, companies can significantly reduce such losses.
Here are some key financial benefits:
- Reduced material waste
- Lower rework and rejection rates
- Improved energy efficiency
- Shorter production cycles
Another major advantage is predictive maintenance. By continuously monitoring Critical Process Parameters (CPP), companies can detect early signs of equipment issues. This allows for timely maintenance, preventing costly breakdowns and downtime.
On the compliance side, regulatory bodies like the FDA and ISO place heavy emphasis on controlling Critical Process Parameters. Companies must demonstrate that their processes are stable, predictable, and well-documented. Failure to do so can lead to penalties, recalls, or even shutdowns.
Optimized CPP management helps with:
- Meeting regulatory requirements
- Simplifying audits and inspections
- Ensuring traceability and documentation
- Reducing risk of non-compliance
There’s also a strategic advantage. Companies that excel in managing Critical Process Parameters often gain a competitive edge. They can produce higher-quality products at lower costs, which is a win-win scenario.
In simple terms, optimizing CPP is like tuning an engine. When everything runs smoothly, you get better performance, lower fuel consumption, and fewer breakdowns. The same principle applies here—efficient control of Critical Process Parameters leads to a more profitable and sustainable operation.
Best Practices for CPP Management
Managing Critical Process Parameters (CPP) effectively requires more than just tools and technology—it demands a disciplined approach and a commitment to continuous improvement. Companies that excel in CPP management don’t treat it as a one-time task; they integrate it into their daily operations and long-term strategy.
One of the most important best practices is documentation. Every Critical Process Parameter should be clearly defined, including its acceptable range, monitoring method, and corrective actions. This ensures consistency across teams and shifts, reducing the risk of errors.
Another key practice is regular review and optimization. Processes evolve over time due to changes in materials, equipment, or production scale. If Critical Process Parameters are not updated accordingly, they can become outdated and less effective.
Here are some essential best practices:
- Establish clear SOPs (Standard Operating Procedures)
- Train employees regularly on CPP importance
- Use real-time monitoring systems
- Conduct periodic audits and reviews
- Implement continuous improvement programs
Collaboration is also crucial. CPP management shouldn’t be limited to one department. Quality assurance, production, engineering, and maintenance teams should work together to ensure that Critical Process Parameters (CPP) are properly controlled.
Another powerful approach is leveraging data. Modern systems generate vast amounts of information about Critical Process Parameters. By analyzing this data, companies can identify trends, optimize processes, and make informed decisions.
Finally, adopting a proactive mindset is essential. Instead of reacting to problems, companies should anticipate them. This means using predictive analytics and risk assessment tools to stay ahead of potential issues.
When these best practices are followed, managing Critical Process Parameters (CPP) becomes not just easier, but significantly more effective.
Future Trends in Critical Process Parameters
The world of Critical Process Parameters (CPP) is evolving rapidly, driven by advancements in technology and increasing demands for efficiency and quality. What once required manual monitoring and basic tools is now becoming a highly automated, data-driven process.
One of the biggest trends is the integration of Artificial Intelligence (AI) and Machine Learning (ML). These technologies can analyze massive datasets to identify patterns and predict deviations in Critical Process Parameters before they occur. This allows companies to move from reactive to predictive process control.
Another emerging trend is the use of digital twins. A digital twin is a virtual replica of a physical process that allows companies to simulate and optimize Critical Process Parameters (CPP) in a risk-free environment. This can significantly reduce the time and cost associated with process development.
Key future trends include:
- AI-driven process optimization
- Real-time predictive analytics
- Cloud-based monitoring systems
- Integration of IoT devices
- Advanced automation and robotics
Sustainability is also becoming a major focus. Companies are using Critical Process Parameters to optimize energy consumption and reduce environmental impact. For example, adjusting parameters to minimize waste or improve efficiency can contribute to greener operations.
Another interesting development is increased regulatory emphasis on data integrity. As systems become more digital, ensuring the accuracy and security of Critical Process Parameters data will be critical.
The future of CPP management is smart, connected, and highly efficient. Companies that embrace these trends will not only improve their processes but also gain a significant competitive advantage.
Conclusion
Critical Process Parameters (CPP) are the backbone of modern process control, bridging the gap between operational efficiency and product quality. By identifying, monitoring, and optimizing these parameters, companies can ensure consistent output, reduce waste, and meet strict regulatory requirements. From pharmaceuticals to manufacturing and food production, the importance of Critical Process Parameters cannot be overstated.
FAQs
1. What are Critical Process Parameters (CPP)?
Critical Process Parameters (CPP) are key variables in a manufacturing process that directly impact product quality and consistency.
2. How are CPP different from CQA?
CPPs are process variables, while CQAs are the final product attributes that define quality.
3. Why are CPP important?
They ensure consistent product quality, reduce defects, and help meet regulatory requirements.
4. How do you identify CPP?
Through risk assessment methods like FMEA and tools like Design of Experiments (DoE).
5. What industries use CPP?
Pharmaceuticals, manufacturing, food processing, biotechnology, and more.