Scale-up studies in pharmaceuticals play a crucial role in transforming laboratory research into full-scale commercial production. In the pharmaceutical industry, developing a drug is only the first step; ensuring that the same process works efficiently on a larger scale is equally important. This is where scale-up studies become essential.
Scale-up studies in pharmaceuticals involve increasing the batch manufacturing size from laboratory or pilot scale to commercial manufacturing while maintaining product quality, safety, and efficacy. These studies help identify potential challenges and ensure that the process remains consistent at every stage. Without proper scale-up, even the most promising drug formulations can fail during production.

What are Scale-Up Studies in Pharmaceuticals?
Scale-up studies in pharmaceuticals refer to the systematic process of increasing production capacity from small-scale laboratory experiments to large-scale manufacturing. The goal is to replicate the same product quality and performance observed during development at a commercial level.
In simple terms, scale-up studies ensure that a process developed in a lab can be successfully implemented in a manufacturing plant. This involves careful evaluation of process parameters, equipment design, and material properties. Any variation during scaling up can affect the final product, making these studies a critical part of pharmaceutical development.
Importance of Scale-Up Studies in Pharmaceuticals
Scale-up studies in pharmaceuticals are essential because they bridge the gap between research and commercial production. Without proper scale-up, there is a high risk of product failure, quality issues, and regulatory non-compliance.
These studies help in identifying critical process parameters that influence product quality. They also ensure that the manufacturing process is robust and reproducible. Additionally, scale-up studies reduce the chances of batch failures, saving both time and cost for pharmaceutical companies. In today’s competitive market, efficient scale-up is key to faster product launch and business success.
Objectives of Scale-Up Studies
The primary objective of scale-up studies in pharmaceuticals is to ensure smooth transition from lab-scale to production-scale manufacturing. However, there are several specific goals associated with these studies.
One major objective is to maintain product quality and consistency across different scales. Another important goal is to optimize process parameters such as temperature, pressure, mixing speed, and time. Scale-up studies also aim to select suitable equipment that can replicate lab conditions at a larger scale. Furthermore, these studies help in identifying potential risks and developing strategies to mitigate them.
Types of Scale-Up in Pharmaceuticals
Scale-up studies in pharmaceuticals can be categorized into different types based on the stage of development.
1. Laboratory to Pilot Scale
This is the first stage of scaling up, where the process is tested in a slightly larger setup. It helps in identifying initial challenges and refining process parameters.
2. Pilot to Commercial Scale
In this stage, the process is transferred to full-scale production. This requires careful planning and validation to ensure consistency and efficiency.
3. Equipment Scale-Up
This involves selecting and designing equipment suitable for large-scale production. Equipment differences can significantly impact process performance.
4. Process Scale-Up
This focuses on maintaining process parameters and ensuring reproducibility at larger scales.
Process of Scale-Up in Pharmaceuticals
The process of scale-up in pharmaceuticals is a systematic and carefully controlled progression that transforms a drug from initial discovery to full-scale commercial manufacturing. Each stage in the scale-up process plays a critical role in ensuring that the product maintains its quality, safety, and efficacy as production volume increases. Understanding this process is essential for successful industrial implementation and regulatory approval.
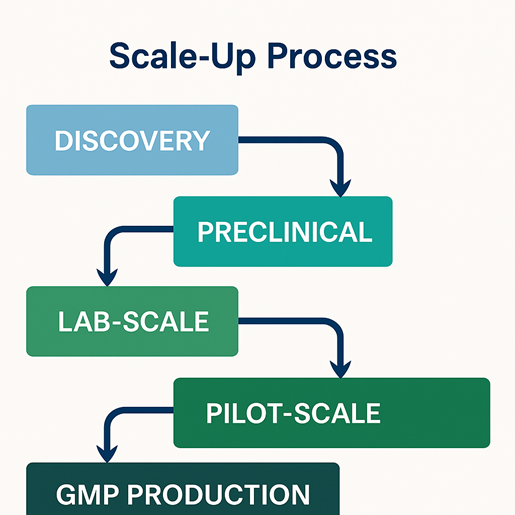
1. Discovery Stage
The scale-up process in pharmaceuticals begins with the discovery stage, where new drug molecules are identified and evaluated for their therapeutic potential. At this stage, research scientists focus on understanding the chemical properties, biological activity, and stability of the compound.
Although production is not the main focus here, early insights into the manufacturability of the drug are important. These initial findings lay the foundation for future scale-up studies in pharmaceuticals by identifying potential challenges in formulation and processing.
2. Preclinical Stage
In the preclinical stage, the drug candidate undergoes laboratory testing and animal studies to evaluate its safety and effectiveness. During this phase, small-scale formulations are developed and tested to determine the best composition and dosage form.
Scale-up studies in pharmaceuticals begin to take shape here, as researchers start considering how the formulation can be produced consistently. Process parameters such as mixing, temperature, and reaction conditions are studied to ensure reproducibility.
3. Lab-Scale Development
Lab-scale development is a critical phase in the scale-up process in pharmaceuticals. At this stage, scientists develop and optimize the manufacturing process on a small scale, typically in laboratory equipment.
The primary goal is to establish a robust and reproducible process that consistently produces high-quality products. Key parameters such as mixing speed, granulation time, drying conditions, and compression force are carefully controlled. These parameters form the basis for further scale-up studies in pharmaceuticals and must be well understood before moving to larger scales.
4. Pilot-Scale Production
Pilot-scale production is the bridge between laboratory development and commercial manufacturing. In this stage, the process is tested in larger equipment that closely resembles industrial systems.
Scale-up studies in pharmaceuticals are most critical at this stage because it helps identify challenges that may not be visible at the lab scale. Factors such as heat transfer, mass transfer, and equipment geometry can significantly impact process performance. Pilot studies allow manufacturers to optimize these parameters and ensure that the process can be successfully scaled up.
5. GMP Production (Commercial Scale)
The final stage in the scale-up process in pharmaceuticals is GMP (Good Manufacturing Practice) production. At this stage, the process is implemented at full commercial scale under strict regulatory guidelines.
Scale-up studies in pharmaceuticals ensure that the product manufactured at this level meets all quality standards and regulatory requirements. Process validation is carried out to confirm that the manufacturing process is consistent and reproducible. Any variation at this stage can lead to batch failures, making it essential to have a well-optimized scale-up strategy.
6. Process Validation and Continuous Monitoring
Even after commercial production begins, the scale-up process in pharmaceuticals does not end. Continuous monitoring and validation are required to maintain product quality and process efficiency.
Advanced tools such as Process Analytical Technology (PAT) and automation systems are used to monitor critical parameters in real time. This ensures that any deviations are detected early and corrected promptly. Ongoing scale-up studies in pharmaceuticals help improve the process and adapt to changing production requirements.
Key Parameters in Scale-Up Studies
Understanding critical parameters is essential for successful scale-up studies in pharmaceuticals.
1. Mixing and Agitation
Proper mixing ensures uniform distribution of ingredients. Scaling up mixing requires careful consideration of impeller design and speed.
2. Heat Transfer
Temperature control becomes more complex at larger scales. Efficient heat transfer systems are required to maintain process stability.
3. Mass Transfer
Mass transfer affects reaction rates and product quality. It must be optimized during scale-up.
4. Reaction Kinetics
Chemical reactions may behave differently at larger volumes, requiring adjustments in process conditions.
5. Equipment Geometry
Differences in equipment size and shape can influence process performance.
Steps Involved in Scale-Up Studies in Pharmaceuticals
Scale-up studies in pharmaceuticals follow a structured approach to ensure successful implementation.
Step 1: Process Understanding
A thorough understanding of the process is essential before scaling up. This includes identifying critical parameters and potential risks.
Step 2: Feasibility Analysis
The feasibility of scaling up the process is evaluated based on technical and economic factors.
Step 3: Pilot Trials
Pilot-scale experiments are conducted to test the process under controlled conditions.
Step 4: Parameter Optimization
Process parameters are optimized to achieve desired product quality.
Step 5: Equipment Selection
Suitable equipment is selected based on process requirements.
Step 6: Validation
The scaled-up process is validated to ensure consistency and compliance with regulatory standards.
Challenges in Scale-Up Studies
Scale-up studies in pharmaceuticals come with several challenges that must be addressed carefully.
One major challenge is maintaining product quality across different scales. Small variations in process parameters can lead to significant changes in product characteristics. Another challenge is equipment differences, as lab-scale equipment may not behave the same way as industrial equipment.
Additionally, heat and mass transfer limitations can affect process efficiency. Regulatory requirements also add complexity, as companies must demonstrate that the scaled-up process meets quality standards. Overcoming these challenges requires expertise and careful planning.
Scale-Up Techniques and Strategies
There are various techniques used in scale-up studies in pharmaceuticals to ensure successful outcomes.
Geometric similarity is one common approach, where the design of larger equipment is kept similar to smaller systems. Another technique is maintaining constant power per unit volume to ensure consistent mixing. Dimensional analysis is also used to study the relationship between different process variables.
Process modeling and simulation tools are increasingly used to predict scale-up performance. These strategies help in minimizing risks and improving efficiency during scaling up.
Role of Quality Assurance in Scale-Up
Quality assurance plays a critical role in scale-up studies in pharmaceuticals. It ensures that the scaled-up process meets regulatory requirements and maintains product quality.
QA teams are responsible for reviewing process changes, approving validation protocols, and ensuring compliance with Good Manufacturing Practices (GMP). They also monitor critical parameters and conduct audits to ensure process consistency. Without proper quality assurance, scale-up efforts may fail to meet industry standards.
Regulatory Considerations
Regulatory compliance is a key aspect of scale-up studies in pharmaceuticals. Regulatory authorities require detailed documentation of the scale-up process, including changes in equipment, process parameters, and validation results.
Companies must demonstrate that the scaled-up process produces products of the same quality as the original formulation. This includes stability studies, process validation, and risk assessment. Meeting regulatory requirements ensures smooth product approval and market entry.
Applications of Scale-Up Studies
Scale-up studies in pharmaceuticals are applied in various areas of drug development and manufacturing.
They are used in the production of tablets, capsules, injectables, and other dosage forms. Scale-up is also important in API manufacturing, where chemical reactions need to be optimized for large-scale production. Additionally, scale-up studies are used in biotechnology processes such as fermentation and cell culture.
Future Trends in Scale-Up Studies
The future of scale-up studies in pharmaceuticals is driven by technological advancements and digital transformation.
Artificial intelligence and machine learning are being used to predict process behavior and optimize parameters. Digital twin technology allows simulation of manufacturing processes, reducing the need for physical trials. Continuous manufacturing is also gaining popularity, reducing the complexity of scale-up.
These innovations are making scale-up studies more efficient and reliable, helping companies bring products to market faster.
Best Practices for Successful Scale-Up
To ensure successful scale-up studies in pharmaceuticals, companies should follow best pharma jobs for practices.
Develop a strong process understanding
Identify critical process parameters early
Use pilot studies effectively
Invest in advanced technologies
Ensure cross-functional collaboration
Maintain proper documentation
Following these practices helps in reducing risks and improving process efficiency.
Conclusion
Scale-up studies in pharmaceuticals are a vital step in the journey from drug development to commercial production. They ensure that processes developed in laboratories can be successfully implemented at an industrial scale without compromising quality.
With proper planning, advanced technologies, and regulatory compliance, scale-up studies can significantly improve production efficiency and reduce risks. As the pharmaceutical industry continues to evolve, scale-up studies will remain a key factor in achieving successful and sustainable manufacturing.
Frequently Asked Questions (FAQ)
1. What are scale-up studies in pharmaceuticals?
Scale-up studies involve increasing production from laboratory scale to commercial scale while maintaining product quality.
2. Why are scale-up studies important?
They ensure consistent product quality and reduce risks during large-scale manufacturing.
3. What are the main challenges in scale-up?
Maintaining quality, equipment differences, and process optimization.
4. What is pilot scale in pharmaceuticals?
It is an intermediate stage between lab and commercial production used for testing processes.
5. What is process validation in scale-up?
It is the process of ensuring that the scaled-up system consistently produces quality products.
6. What are critical process parameters?
These are variables that impact product quality and must be controlled during manufacturing.
7. How does scale-up affect product quality?
Improper scaling can lead to variations in product characteristics.
8. What technologies are used in scale-up studies?
Simulation tools, AI, and process analytical technology.